Conductivity in Water Chemistry and Molecular Science
Conductivity is a basic water quality measure. It shows how well water can carry an electrical current. It serves as a practical indicator of the dissolved substances within a water sample or body of water. High conductivity levels indicate many dissolved contaminants, while lower levels suggest cleaner water with fewer impurities.
This parameter is widely used to measure ion levels in water solutions. It covers many dissolved compounds. Conductivity sensors are often used to measure solute levels in solutions and to monitor water purification processes.
In water chemistry and molecular science, conductivity plays a key role. It tells industries and city systems about water quality. It helps decide if the water is safe to drink or needs treatment. Among water quality measures, conductivity can show if water is clean or may be contaminated.
Conductivity is typically expressed in terms of micro-Siemens per centimeter (µS/cm) or milli-Siemens per centimeter (mS/cm). Establishing baseline readings helps you monitor water quality over time. It ensures you maintain acceptable conductivity levels. This article delves deeper into the concept of conductivity and its significance in water chemistry.
Electrical Conductivity and Water
Water and electricity are rarely seen as safe together. For instance, water near an electrical outlet can make the socket stop working. It can also cause sparks and fire hazards.
However, pure water, with no contaminants, is a strong insulator.
This means it does not conduct electricity. Without impurities, there are no free ions to help conduct electricity.
That said, pure water is rare. Water tends to dissolve almost anything it touches. Whether drawn from a kitchen faucet or filled into a swimming pool, it typically contains dissolved minerals, chemicals, and other substances.
These solutes enhance conductivity, as the presence of dissolved ions increases a solution’s ability to conduct electrical currents. By filtering out these impurities, conductivity levels can be greatly reduced.
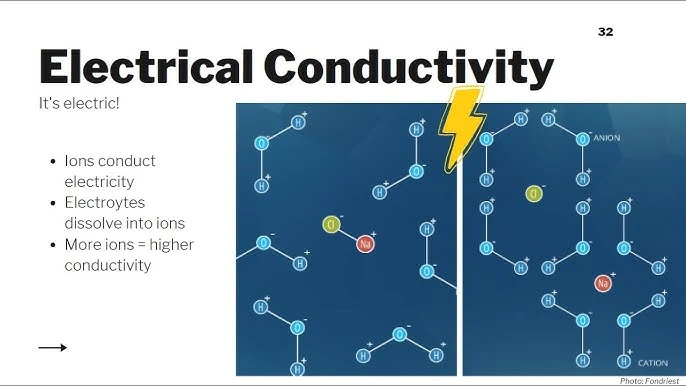
The Role of Free Ions in Conducting Electricity
Once substances dissolve in water, the water becomes less insulating. Dissolution creates free ions, usually from salts. Salts are ionic compounds with positively charged cations and negatively charged anions. In water, these ions usually balance each other, making the solution electrically neutral.
Even at low ion levels, water can still conduct electricity. For example, adding salt to bathwater during a thunderstorm can raise the chance.
The electrically charged lightning could then interact with ions in the water. Electricity naturally moves toward opposite charges, which helps it travel through water.
When water contains high levels of ions and solutes, it becomes an efficient conductor. An electrical current passing through such a solution may disregard other contents in the water. In fact, a person standing in highly ionized seawater may have a lower risk of electrocution during lightning.
This risk may be lower than in less conductive environments. This explains why the risk of electrocution at sea is usually low.
Is Conductivity a Physical or Chemical Property?
Before measuring conductivity, it helps to know if it is a physical or chemical property. A **physical property** is an attribute you can observe or measure. It does not change a substance’s chemical structure. Examples of physical properties include:
– Molecular weight
– Volume
– Color
– Hardness
– Density
– Melting point
– Boiling point
In contrast, **chemical properties** need changes in a substance’s chemical makeup to be observed. This often involves chemical reactions. Common examples include:
– Flammability
– Reactivity
– Acidity
– Toxicity
– Heat of combustion
Electrical conductivity is a physical property. It does not change the substance’s chemical identity. The process only transfers energy, such as heat or electricity, without changing molecular structure or composition. Thus, it is a non-invasive method to assess key characteristics of solutions.
How Conductivity Impacts Water Quality
Conductivity is key for checking water quality. It measures the amount of dissolved salts in a water sample. Higher conductivity levels often show inorganic chemicals are present. This reflects how salty the water is.
Higher salt levels raise conductivity readings and can also signal possible contamination. As a result, conductivity is a reliable way to measure water quality.
Measuring Conductivity in Water
To measure water conductivity, conductance testing is done using conductivity sensors. This technique measures how well water conducts electric current, which relates to ion levels and movement. These ions form when compounds dissolve. They carry positive or negative charges and allow electricity to flow.
Factors Indicating Poor Water Quality Through Conductivity Changes
Many factors can cause changes in conductivity readings. Significant changes often suggest lower water quality, as more pollution raises conductivity and worsens water standards.
Salinity
Salinity is one of the main causes of water conductivity. Pure water with no salt cannot conduct electricity. But even small amounts of salt cause a clear rise in conductivity. While salinity and conductivity are related, conductivity is easier to measure. It is often used to estimate salinity levels.
Total Dissolved Solids (TDS)
Solida dissoluta tota sunt salia, metalla, et mineralia in aqua dissoluta.
Saepissime sunt salia inorganica, ut magnesium, natrium, kalium, calcium, chlorida, sulfata, et bicarbonata. High TDS levels can disrupt cell functions in aquatic organisms. This can affect how they move in the water. It may cause abnormal floating or sinking.
Temperature
Temperature affects conductivity. Warm water causes higher readings. Cool water causes lower readings.
The Importance of Monitoring Conductivity to Protect Water Quality
Regularly checking water conductivity is vital to protect water quality. Conductivity sensors give precise data on total dissolved solids and chemical levels in water. If conductivity readings increase, it’s likely that impurities are compromising the water’s quality.
Inaccurate measurements could lead to unintentional use of contaminated water that does not meet required standards for cleanliness. Conductivity monitoring helps detect hazards early and fix them fast.
Types of Conductivity Sensors
There are two common types of conductivity sensors: contacting sensors and toroidal sensors. Both provide reliable readings, but toroidal sensors work best in very dirty water. Even when covered with dirt or other pollutants, these sensors still work due to their robust design.
Toroidal sensors function through:
– A continuous waveform is produced inside a transmission coil.
– An alternating magnetic field in the coil creates voltage in the medium.
– Free ions create alternating current as anions and cations move within the medium.
– The resulting current creates a secondary current in the receiving coil.
– Higher ion levels increase conductivity and current strength.
Monitoring conductivity at home or in city facilities is an effective way to check for water contaminants. High readings often mean there are many impurities. Low readings suggest the water is clean and well filtered.