What pH Represents and Why It’s the Benchmark for Modern Sensors
The concept of pH is key in many industries, from food production to environmental monitoring. But what does pH really mean? Why do modern sensors use it so often? This article explains what pH means and why it still matters in science and industry.
Understanding the Basics of pH
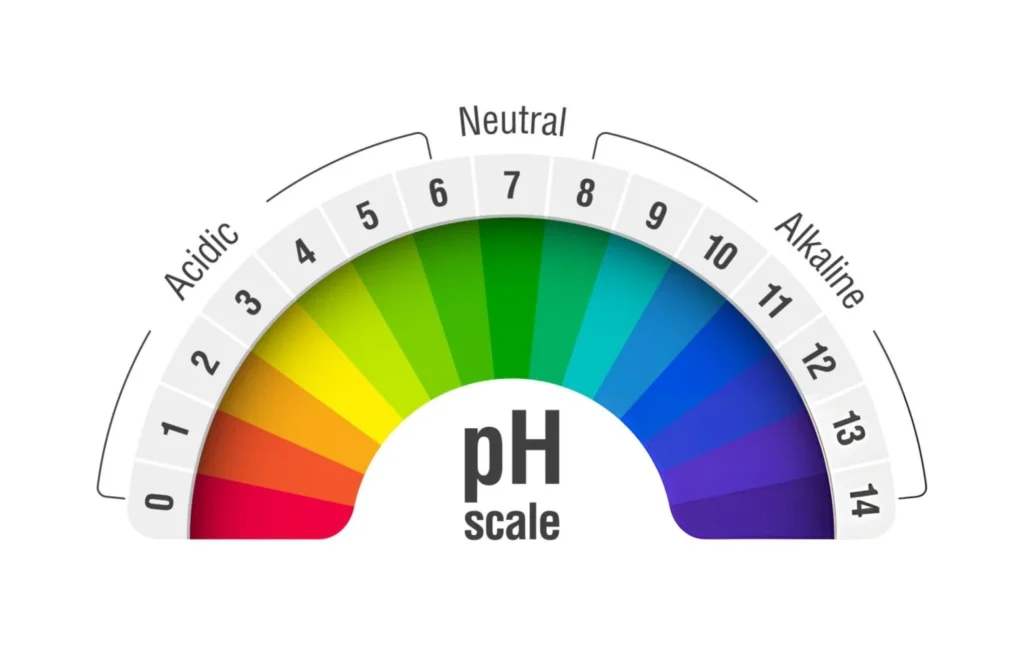
pH means “potential of hydrogen.” It measures the amount of hydrogen ions (H⁺) in a solution. The pH scale ranges from 0 to 14. A pH of 7 is neutral, like pure water. Values below 7 mean acidity. Values above 7 mean alkalinity, also called basicity.
The scale is logarithmic, so each unit shows a ten times change in acidity or alkalinity. For example, a solution with a pH of 4 is ten times more acidic than one with a pH of 5. This is important in chemical analysis.
The Science Behind pH
The pH scale shows the balance between acids and bases in a solution. Acids release hydrogen ions (H⁺), which raises acidity.
Bases lower H⁺ levels by producing hydroxide ions (OH⁻). Water (H₂O) naturally splits into H⁺ and OH⁻ ions. This makes it key to measuring pH.
The pH measurement thus reflects this ionic balance in various liquids, helping to predict chemical behavior under different conditions.
Why pH is a Key Metric for Sensors
Among measurable factors, pH is a strong choice for sensors for several clear reasons:
– **Quantitative Precision**:
The pH scale gives steady, exact measures for many kinds of solutions.
– **Functional Accuracy**:
Modern pH sensors use a pH-sensitive glass electrode and a reference electrode. They detect voltage differences to give precise readings.
– **Wide Application Range**:
pH sensors are used in many fields, from aquariums to biotech labs. They help keep conditions ideal. They also support quality control.
Advantages of Using pH as a Metric
The use of pH as a sensing measure offers distinct benefits:
1. **High Sensitivity**:
Advanced sensors can detect small changes in hydrogen ion concentration. This enables accurate readings, even in complex solutions.
2. **Real-Time Responsiveness**:
Applications like fermentation or water treatment need constant monitoring. Quick feedback from pH sensors helps operators make fast changes and keep efficiency high.
3. **Global Standardization**:
The pH scale is used worldwide, so measurements can be compared. This helps keep scientific and industrial work consistent.
Challenges in pH Sensing
Despite its benefits, measuring pH can still be challenging:
– **Impact of External Factors**: Temperature shifts, ionic strength changes, and contamination can change readings. This shows why controlled conditions matter.
– **Calibration Requirements**: Regular calibration with buffer solutions keeps the sensor accurate over time.
– **Equipment Lifespan**: Proper maintenance helps sensors work longer and lowers replacement costs.
The Historical Significance of pH
The concept of pH dates back to 1909. Danish biochemist Søren Peder Lauritz Sørensen helped establish it. Søren created the pH scale and changed chemistry. It gave a clear way to measure acids and bases. This made it vital for science and industry.
Contribution to Chemistry
In chemistry, pH is key for studying acid-base reactions. It also helps keep experiments balanced. Its role extends from titration procedures to broader applications like synthesizing products and optimizing reaction kinetics. By showing if a solution is acidic, neutral, or alkaline, pH affects chemical processes and results.
How is pH Measured?
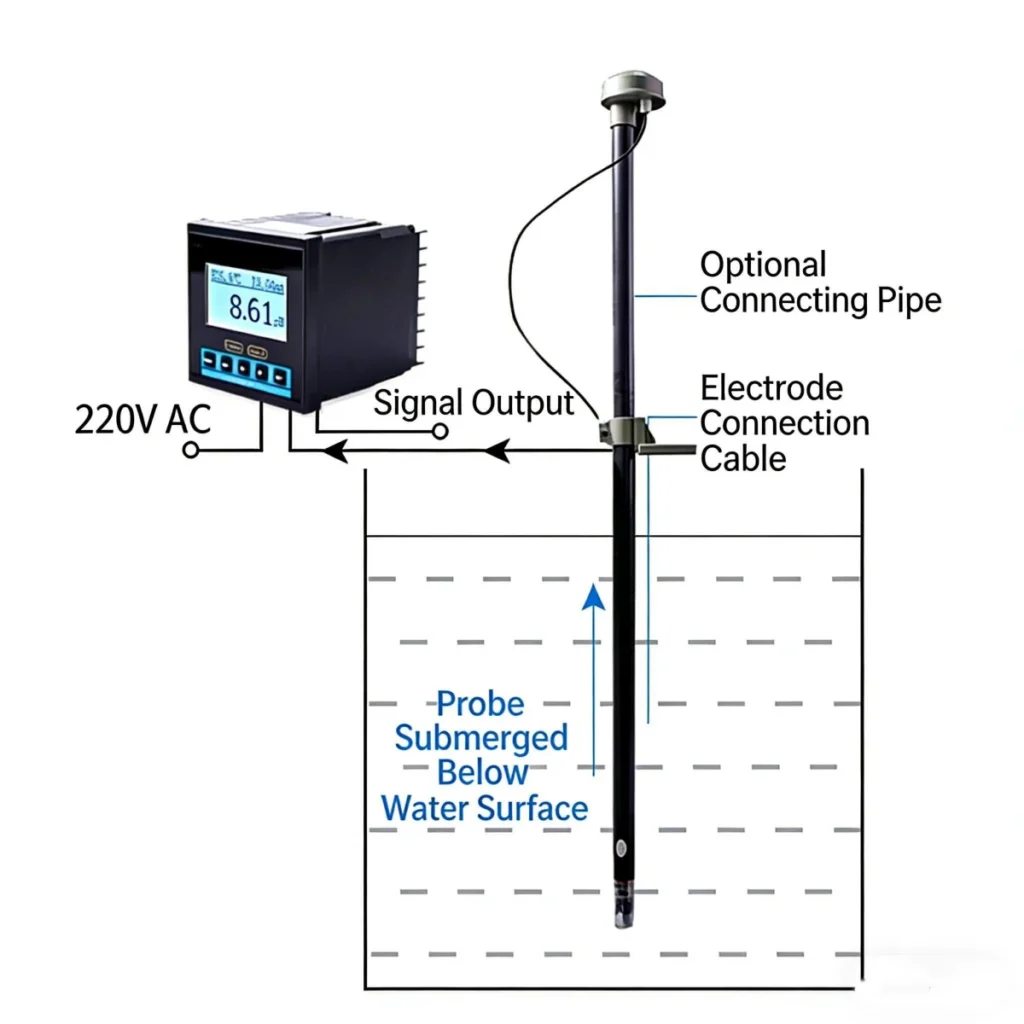
To measure pH, the activity of hydrogen ions (H⁺) in a solution is quantified using sensors or electrodes. A typical setup includes a glass electrode sensitive to H⁺ ions paired with a reference electrode. When placed in liquid, the glass membrane creates a voltage.
This voltage depends on hydrogen ion activity. This voltage matches the solution’s pH value on the 0–14 scale. It is shown on a transmitter or meter.
For accurate results, sensors need regular calibration with buffer solutions and proper care. Temperature changes, contaminants, and aging electrodes can affect measurement reliability.
Standard pH Ranges
Understanding the usual pH range of common substances gives helpful insight into how pH affects daily life.
Highly acidic solutions, like lemon juice and stomach acid, often have a pH near 2. This shows they are strongly acidic. On the other hand, baking soda is a useful household base.
It is used for cooking, cleaning, and as an antacid. It has an alkaline pH of about 9.
Such examples show the wide range of pH values we see each day.
Applications of pH sensors
pH sensors are used far beyond labs. They play a key role in many industries. These include pharmaceuticals, agriculture, food and drink production, and wastewater treatment.
They ensure product quality and safety. And they improve chemical processes. They help meet environmental regulations.
In agriculture, monitoring soil pH helps create the best conditions for crop growth. Meanwhile, in pharmaceuticals, pH sensors help with drug formulation and keeping quality standards.
pH Sensor Technology
pH sensors function on the principles of electrochemistry, comprising a pH-sensitive glass electrode and a reference electrode. When placed in a solution, the pH-sensitive electrode creates a voltage that shows the solution’s pH.
This process uses a selective exchange of hydrogen ions between the solution and the electrode. This exchange enables accurate and reliable measurements.
pH Buffer Solutions
pH buffer solutions are vital for accurate pH measurements. These solutions have stable, known pH values. They serve as benchmarks for calibrating sensors.
Regular calibration with buffer solutions reduces sensor drift and performance changes. It ensures accurate and consistent measurements over time.
Advanced pH Sensing
Recent advances in pH sensor technology have led to new sensors with better features. Features like wireless connectivity, real-time monitoring, and links to automation systems boost process efficiency.
This is true in industries like water treatment and biotech research. These improvements enable faster responses to pH changes and support remote monitoring.
Real-world Examples
Practical uses of pH sensing span many sectors. In the food industry, pH monitoring helps ensure consistent data and product safety for dairy, beer, and soft drinks.
In environmental science, sensors measure water pH in rivers and oceans. They assess aquatic ecosystem health and track changes like ocean acidification. Precise pH measurement is useful for solving global challenges.
Environmental Monitoring
In environmental studies, pH sensing is vital for measuring how human activities affect ecosystems. Tracking water pH in rivers, lakes, and oceans helps scientists watch trends like ocean acidification.
Rising carbon dioxide levels in the air can cause this change. This key data can guide choices about water quality management and protecting aquatic habitats.
Future Trends
Ongoing advances in sensor technology point to exciting new possibilities for the future of pH measurement. For instance, wearable health devices with pH sensors may let people monitor their body’s pH levels for wellness.
In addition, the growing Internet of Things ecosystem may use real-time data from pH sensors. It can improve decision-making in sectors from agriculture to healthcare.
Conclusion: The Importance of pH Sensing Across Time
As pH sensing technology evolves, designs become more accurate, reliable, and versatile for many needs. The growing recognition of its value across new industries ensures it stays relevant well into the future.
Whether you study chemical reactions, improve industry processes, or just wonder about pH, this universal measure matters. It has a strong impact on science and industry.